Build Smarter Gut Health Products for Europe
European gut health markets are evolving rapidly—but so are the scientific and regulatory expectations behind “biotics.” In practice, choosing between prebiotics, probiotics, and postbiotics is no longer just a formulation decision; rather, it is a strategic choice that affects claims, stability, logistics, and market access.
If you are planning a new gut health product for Europe, then consider partnering with Klee to access science-backed, formulation-ready ingredients designed for EU compliance and scalability.
Scientific Definitions and Clear Terminology
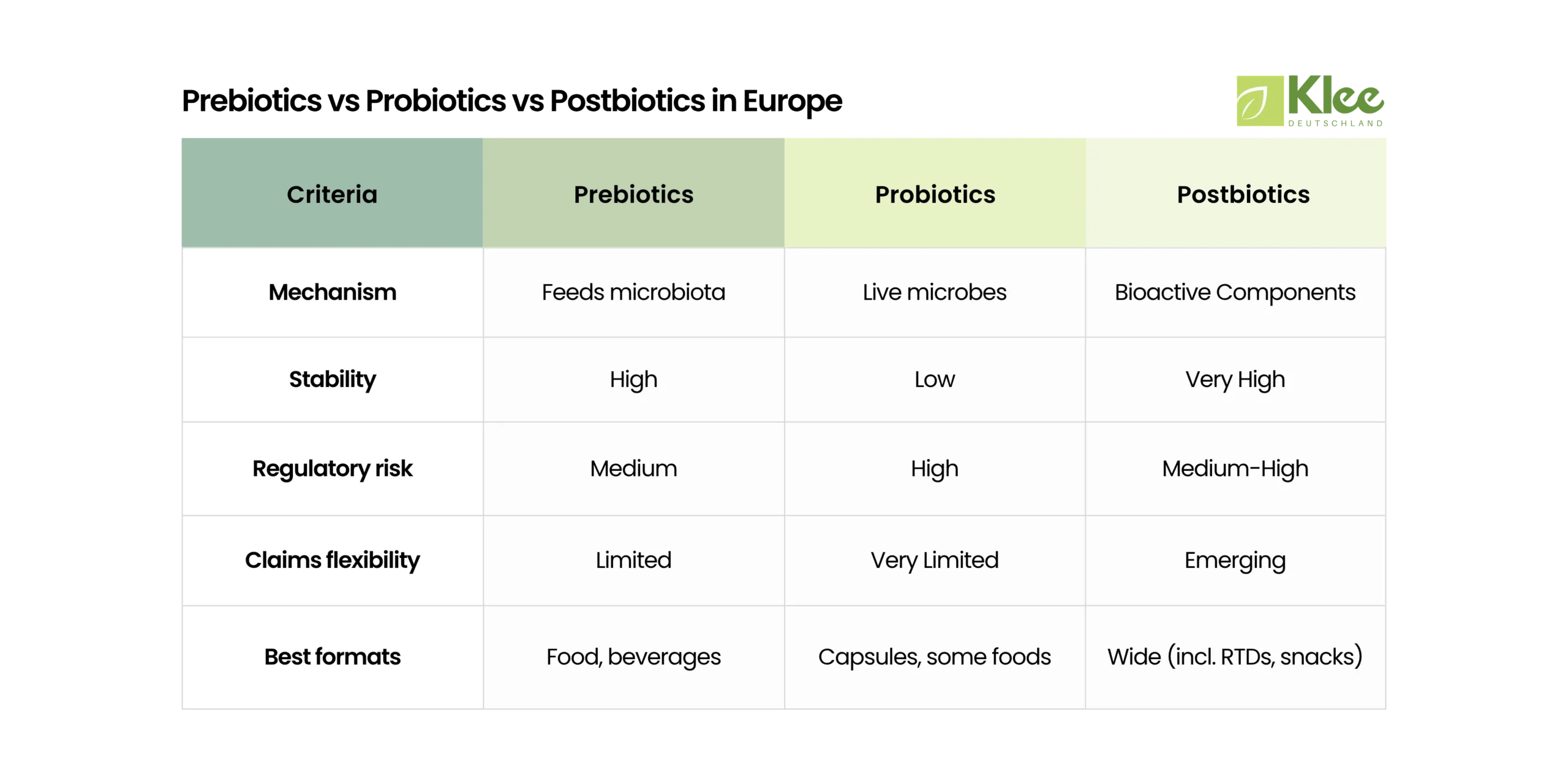
First, a precise understanding of terminology is essential for B2B brands operating in Europe. In particular, the terms “prebiotics,” “probiotics,” and “postbiotics” are not interchangeable—they reflect fundamentally different scientific concepts with distinct regulatory implications.
Prebiotics
The International Scientific Association for Probiotics and Prebiotics (ISAPP) defines prebiotics as:
“a substrate that is selectively utilized by host microorganisms conferring a health benefit.”
This definition emphasizes:
- selectivity (targeting beneficial microbes),
- mechanistic relevance, and
- demonstrated health outcomes.
For example, typical prebiotic ingredients include inulin, fructo-oligosaccharides (FOS), and galacto-oligosaccharides (GOS).
Probiotics
Probiotics are defined by FAO/WHO as:
“live microorganisms which when administered in adequate amounts confer a health benefit on the host.”
This definition introduces strict requirements:
- viability at time of consumption,
- strain-level identification,
- demonstrated clinical benefit at the delivered dose.
In Europe, probiotic strains are often assessed using EFSA frameworks, including Qualified Presumption of Safety (QPS).
Postbiotics
ISAPP’s 2021 consensus defines postbiotics as:
“a preparation of inanimate microorganisms and/or their components that confers a health benefit on the host.”
Key clarifications:
- Postbiotics include inactivated microbial cells or cell components.
- Purified metabolites alone do not qualify.
- Health benefits must still be demonstrated through evidence.
Why Definitions Matter for European Brands
In the EU, terminology is not just scientific—it directly impacts:
- health claim eligibility,
- labeling compliance,
- regulatory classification (food vs. novel food).
As a result, using incorrect or unsupported terminology can lead to rejected claims or restricted market access.

Mechanistic Differences in the Gut
Therefore, understanding mechanisms is essential for designing targeted, differentiated products.
Prebiotics: Ecosystem Modulation
Prebiotics act indirectly by:
- feeding beneficial gut microbes,
- increasing production of short-chain fatty acids (SCFAs),
- supporting gut barrier function and immune modulation.
Their effect depends heavily on existing microbiota composition.
Probiotics: Live Microbial Interaction
Probiotics function through:
- transient colonization,
- competitive exclusion of pathogens,
- immune signaling and metabolic activity.
Importantly, many probiotic effects occur without permanent colonization, acting during gastrointestinal transit.
Postbiotics: Direct Bioactive Effects
Postbiotics act as defined bioactive compounds, including:
- microbial cell wall components,
- signaling molecules,
- fermentation-derived compounds within the preparation.
They interact directly with host systems such as:
- immune receptors,
- epithelial cells,
- metabolic pathways.
B2B Insight
- Prebiotics = indirect modulation
- Probiotics = live interaction
- Postbiotics = direct bioactivity
This distinction allows brands to align ingredients with specific product goals, such as:
- daily digestive balance,
- immune targeting,
- precision formulations.
Stability and Shelf-Life Comparison
Importantly, stability is one of the most critical differentiators for commercial success.
Probiotics: High Sensitivity
Probiotic viability is affected by:
- temperature,
- humidity,
- oxygen exposure,
- processing conditions.
Maintaining labeled CFU counts until the end of shelf life is a major technical challenge and regulatory expectation.
Prebiotics: High Chemical Stability
Prebiotics are generally:
- heat stable,
- compatible with standard food processing,
- suitable for a wide range of formats.
However, degradation can occur under:
- extreme pH,
- prolonged heat,
- Maillard reactions.
Postbiotics: Maximum Robustness
Postbiotics eliminate viability concerns entirely:
- no live cells → no survival requirement,
- more stable across processing conditions,
- often compatible with ambient storage.
Because they are non-viable, postbiotics can offer:
- longer shelf life,
- reduced cold-chain dependency,
- greater formulation flexibility.

Practical Implications for Brands
| Factor | Prebiotics | Probiotics | Postbiotics |
| Heat stability | High | Low | High |
| Shelf life | Long | Sensitive | Long |
| Cold chain | Not required | Often required | Rarely required |
| Format flexibility | High | Limited | Very high |
If you are looking to develop shelf-stable gut health products, then Klee’s fermentation-derived ingredients can help you overcome probiotic stability limitations.
Manufacturing and Storage Implications
Prebiotics
Production methods include:
- plant extraction,
- enzymatic synthesis,
- controlled fermentation.
Key B2B considerations:
- purity,
- degree of polymerization,
- consistency across batches.
Probiotics
Manufacturing requires:
- controlled fermentation,
- downstream processing (freeze-drying),
- strict strain documentation.
Critical requirements:
- genomic identification,
- viability validation,
- stability testing.
Postbiotics
Postbiotic production involves:
- fermentation,
- controlled inactivation (heat, pressure),
- validation of retained functionality.
Unlike probiotics:
- viability is not required,
- safety assessment may be simpler in some cases.
Storage and Logistics Strategy
- Probiotics: require tight environmental control
- Prebiotics: stable under ambient conditions
- Postbiotics: ideal for global, flexible distribution
For European brands, this translates into:
- different cost structures,
- varying supply chain risks,
- different scalability potential.

Regulatory Landscape in Europe
From a strategic perspective, the EU regulatory framework is one of the most critical considerations for B2B decision-making.
Health Claims Regulation
Under Regulation (EC) No 1924/2006:
- all health claims must be authorized and scientifically substantiated,
- EFSA evaluates evidence,
- only approved claims can be used.
Within this regulatory framework, the EU Register of Nutrition and Health Claims provides the official list of authorized and rejected claims.
Probiotics: No Approved Health Claims
To date:
- no probiotic health claims have been authorized in the EU,
- the term “probiotic” is often considered an implied health claim.
This creates significant communication challenges for brands.
Prebiotics: Limited Approved Claims
Prebiotic-related claims exist, but:
- they are tied to specific ingredients and effects (e.g., fiber-related benefits),
- the term “prebiotic” itself is not freely usable without substantiation.
Postbiotics: Emerging Category
Postbiotics are not a distinct regulatory category:
- assessed under existing frameworks,
- may fall under novel food regulation depending on history of use.
Key Regulatory Challenges
- inconsistent interpretation across Member States,
- strict evidence requirements,
- terminology restrictions,
- evolving guidance for new categories like postbiotics.
B2B Strategy for Compliance
Successful European brands:
- align early with EFSA expectations,
- design claims around approved physiological effects,
- avoid risky terminology where necessary,
- build strong scientific dossiers.

Commercial and Positioning Implications for Brands
Prebiotics: Scalable and Accessible
Best suited for:
- mainstream food & beverage,
- daily wellness positioning,
- cost-efficient formulations.
Advantages:
- easy integration,
- wide consumer acceptance,
- relatively low regulatory risk.
Probiotics: Premium and Clinical
Best suited for:
- supplements,
- practitioner channels,
- targeted health benefits.
Challenges:
- claims restrictions,
- stability constraints,
- higher cost of goods.
Postbiotics: Innovation and Flexibility
Best suited for:
- shelf-stable formats,
- modern lifestyle products,
- cross-category innovation (e.g., beauty, energy).
Advantages:
- strong stability profile,
- fewer logistical constraints,
- emerging differentiation opportunity.
Strategic Positioning Framework
| Brand Type | Best Fit |
| Mass-market food brands | Prebiotics |
| Clinical/niche supplements | Probiotics |
| Innovative lifestyle brands | Postbiotics |
Want to differentiate in crowded EU markets? Klee supports brands with next-generation gut health ingredients, including fermentation-derived solutions tailored for stability and compliance.
Choosing the Right Strategy Based on Product Goals
The optimal choice depends on clear product objectives.
Choose Prebiotics if:
- you need scalability and cost efficiency,
- your product is food or beverage-based,
- your positioning is daily digestive wellness.
Pick Probiotics if:
- you have strong strain-specific evidence,
- your target is clinical or specialized applications,
- you can manage stability and regulatory complexity.
Choose Postbiotics if:
- you need shelf-stable, flexible formats,
- your product faces processing challenges,
- you want innovation-driven positioning.
Conclusion: A Strategic Decision, Not Just an Ingredient Choice
Overall, the prebiotics probiotics postbiotics comparison for Europe highlights a central truth:
namely, there is no universally superior option—only better strategic fits.
For B2B brands, success depends on aligning:
- scientific mechanism,
- formulation feasibility,
- regulatory compliance,
- commercial positioning.
Those who integrate these dimensions early will:
- reduce development risk,
- accelerate time-to-market,
- build more defensible product concepts.
If you are ready to develop your next-generation gut health product for Europe, then consider partnering with Klee to access stable, compliant, and innovative ingredients tailored to your product goals.
References
European Commission. (n.d.). EU register of nutrition and health claims. Retrieved from EU Register of Health Claims
Food Compliance International. (2023). EU postbiotics: The concept and their use in healthy population. Retrieved from https://foodcomplianceinternational.com/industry-insight/scholarly-articles/3019-eu-postbiotics-the-concept-and-their-use-in-healthy-population
Food and Agriculture Organization of the United Nations & World Health Organization. (2002). Guidelines for the evaluation of probiotics in food.
Salminen, S., Collado, M. C., Endo, A., Hill, C., Lebeer, S., Quigley, E. M. M., et al. (2021). The International Scientific Association of Probiotics and Prebiotics consensus statement on the definition and scope of postbiotics. Nature Reviews Gastroenterology & Hepatology. https://isappscience.org/resource/expert-consensus-document-the-international-scientific-association-for-probiotics-and-prebiotics-isapp-consensus-statement-on-the-definition-and-scope-of-postbiotics
International Scientific Association for Probiotics and Prebiotics (ISAPP). (2023). Frequently asked questions about the ISAPP postbiotic definition. https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2023.1324565
ISAPP. (2017). Consensus statement on the definition and scope of prebiotics. Nature Reviews Gastroenterology & Hepatology.

